Listeria monocytogenes survival in refrigerator dill pickles
Jin Kyung Kim*, Elaine M. D'Sa**, Mark A. Harrison*, Judy A. Harrison**, and Elizabeth L. Andress**
* Dept. of Food Science and Technology, University of Georgia, Athens
** Dept. of Foods and Nutrition, University of Georgia, Athens
Paper 33C-1. Presented at the Institute of Food Technologists Annual Meeting, Las Vegas, NV, July 14, 2004.
Note: This research study analyzed one particular pickling procedure that started with partially fermenting cucumbers at room temperature and then storing them in the refrigerator with no further treatment or processing. It does not represent findings or advice for any other type of refrigerator pickles.
Abstract
Listeria monocytogenes can survive and grow in refrigerated foods with pH levels of approx.
4.0-5.0 and salt concentrations of 3-4%. Home-fermented refrigerator dill pickles fit this
description. Contamination of this product with L. monocytogenes could cause serious problems
since these items are not heated prior to consumption. This study determined L. monocytogenes
survival and growth patterns in refrigerator dill pickles at three salt levels. Pickling
cucumbers were inoculated with L. monocytogenes, brine mixtures were added and the cucumbers
were held at room temperature for one week and then refrigerated for up to 3 months. The pH,
percent NaCl, percent titratable acidity and total aerobic, psychrotrophic, lactic acid
bacteria and Listeria populations were measured at the addition of brine, at 2, 4, and 7
days during storage at room temperature and then at weekly intervals during refrigerated
storage. There was a rapid decrease in pickle pH after four days at room temperature (from
6.1-6.2 to 4.4-4.6) followed by a gradual decrease. The percent NaCl in the pickles rapidly
increased up to 3 weeks at refrigeration temperatures, and the percent titratable acidity
in the highest salt level was significantly lower (p<0.05). The initial Listeria population
was 5.4-5.6 log cfu/cm² on the surface and 3.9-4.6 log cfu/g internally. There was
approximately a 0.5-1 log increase during fermentation at room temperature followed by
a population decline during refrigerator storage, with a greater decrease in the pickles
with the highest NaCl content. Populations of total aerobes and lactic acid bacteria
increased during room temperature storage and decreased gradually.
Introduction
-
L. monocytogenes has been a major food safety concern since the
1st outbreak due to consumption of coleslaw in 1981 (Shlech 1983)
- L. monocytogenes is widely distributed in the environment
- Isolated from various environment and food sources
- Cross-contamination with L. monocytogenes can occur
- Found within the food manufacturing environment
- Can attach to various manufacturing surfaces and produce a biofilm
-
Adaptation of L. monocytogenes to environmental stresses
- pH: can survive and/or grow as low as pH 4.4 (George 1988)
- Salt concentration: can survive up to 14% NaCl (Farber 1992)
- Temperature: remain metabolically active at 3°C (Walker 1990)
-
Pickling (Brandt 1996)
- Preserving vegetables, meat and fish with salt and acid
- Cucumbers are one of the more commonly pickled foods in the U.S.
-
Home-fermented refrigerator dill pickles
- Lactic acid fermentation of cucumbers
- Made by immersing the pickling cucumbers into a brine solution
- Kept at room temperature for 1 week followed by storage at refrigeration temperatures during the consumption period
Objective
To determine the fate of L. monocytogenes on the surface and in the interior of cucumbers and in brines
subjected to the different salt levels (1.3, 3.8, 7.6%)
Materials and Methods
- Bacterial strains and inoculum preparation
- Five L. monocytogenes strains, 301, V7, LCDC, Scott A, and Brie used
- Cultured in 10 ml of TSB and incubated for 24 h at 37°C
- Transferred to 4,000 ml of TSB and incubated for 24 h at 37°C
- Centrifuged at 4,550 x g for 30 min
- Pellet was resuspended into sterile 0.1% peptone water
- Inoculation of cucumbers
- Long, unwashed, pickling cucumbers (approx. 10 cm)
- Washed with tap water and drained for 30 min
- Immersed cucumbers into an inoculum of L. monocytogenes for 15 min
- Cucumbers drained on a sterile metal grid rack for 15 min
- 3 salt levels (1.3, 3.8, and 7.6%) with L. monocytogenes and a control treatment of 3.8% NaCl and no inoculum used
- Physical Analyses
- pH measurement in cucumbers and brines with a pH meter
- Percent titratable acidity: titrated 20 g of sample with 0.1N NaOH - % Acid = (mL NaOH)(N NaOH)(milieq. wt of acid)(100)(wt of sample(g))-1
- Percent NaCl: Quantab chloride titration strips with calibration table
- Microbiological properties
- Sampling interval: at the addition of brine (0), 2, 4, 7 d at room temperature, 1, 2, 3, 4, 6, 8, 10, 12 weeks during refrigerated storage
-
Sampling procedure
- Surface of cucumber: cut into 2.5 x 2.5 cm pieces using sterile knife
- Interior of cucumber:10 g, using sterile corer from the blossom end
- Brine: directly diluted using peptone water
-
Microbiological count
- Total aeobes: PCA, incubated at 35°C for 24 h
- Psychrotrophs : PCA, incubated at 7°C for 7 d
- Lactic acid bacteria: MRS 2X, incubated at 35°C for 48 h anaerobically
- Listeria: LSA (Listeria Selective Agar), incubated at 35°C for 48 h
-
Listeria enrichment
- 1 ml of from bag inoculated into 9 ml LEB (Listeria Enrichment Broth)
- Incubated at 30°C for 48 h
- Streaked onto LSA and incubated at 35°C for 48 h
- Checked for Listeria colonies
-
Identification and confirmation test
- Gram stain, Catalase/Oxidase reaction
- Umbrella-like growth in motility medium
- Biochemical testing using Micro ID kits (Remel, Lenex, KS)
- Statistical Analysis
- ANOVA (Statistical Analysis Systems Institute, Cary, NC)
- Duncan's multiple range tests - significance value:
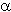 =0.05
=0.05
Results
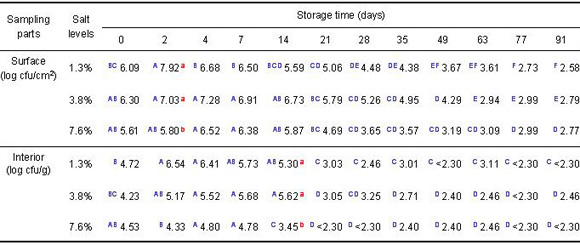
Table 1. Population of
L. monocytogenes on surface and in interior of cucumbers at three salt levels

1/3 Number of tubes showing positive result /number of replicates
- Initial population - Surface: 5.4–5.6 log cfu/cm²; Interior: 3.9–4.6 log cfu/g
- Listeria populations decreased the most in the highest salt content during refrigerator storage
-
Listeria populations were not detected on surfaces or in the interiors, after 8
and 4 weeks at refrigerator temperature, respectively
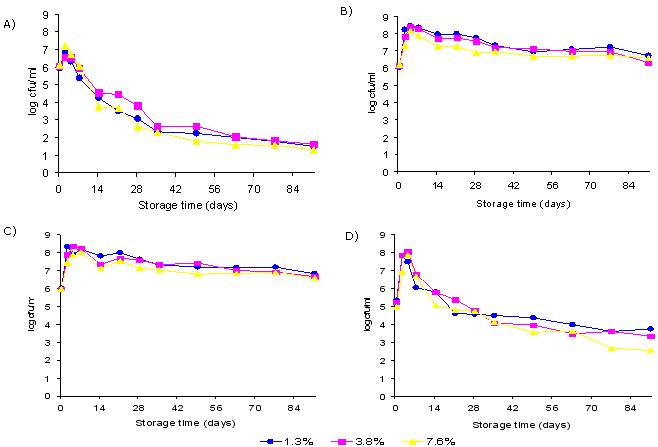
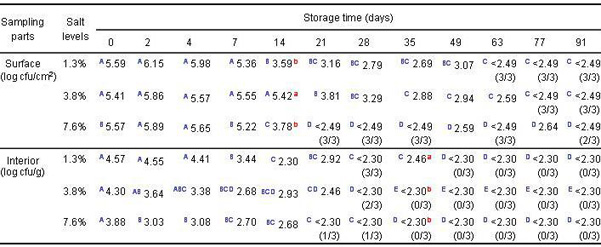
Fig. 1. Change patterns of microorganisms in the brines at three salt levels
A)Listeria B) Total Aerobes C) Lactic Acid Bacteria D) Psychrotrophs
- Similar change patterns were observed for each type of microorganism
- Initial population of L. monocytogenes: 6–6.1 log cfu/ml
- Total aerobes increased by 3 logs after 4 d at room temperature and then, gradually decreased
- No significant difference (p>0.05) in the number of lactic acid bacteria among salt levels
Table 2. Population of total aerobes on surface and in interior of cucumbers at three salt levels

- Aerobic populations were significantly higher (p<0.05) at 1.3 % salt levels
- Aerobic populations were greatest at 4 (surface) or 7 (interior) d at room temperature
- Aerobic populations decreased gradually during refrigeration temperature
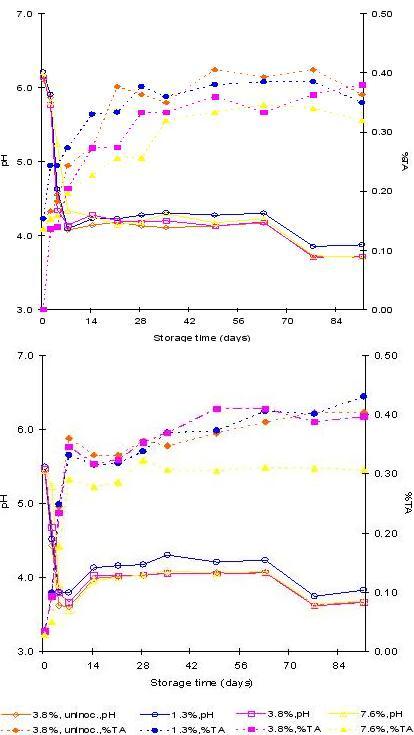
Fig. 2. The pH and percent titratable acidity at three salt levels - A) Cucumbers B) Brines
-
There was a rapid pH decrease from 6.1–6.2 to 4.4–4.6 (cucumbers), from 5.5 to 3.6–3.9
(brines), except the highest salt level (from 6.2 to 5.2), by the 4th day.
- Significantly lower (p<0.05) percent titratable acidity for brines in 7.6% salt level
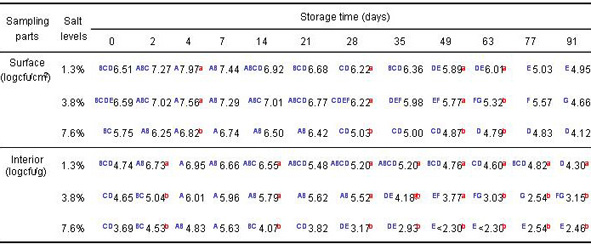
Table 3. Population of psychrotrophs on surface and in interior of cucumbers at three salt levels

- Psychrotrophic populations increased 1-2 logs at room temperature and then decreased
- Psychrotrophic populations decreased 2-3.5 logs at the end of storage time
- For each sampling day there was no significant difference (p<0.05) in psychrotrophic populations in the pickle interiors among salt levels
Conclusion
-
Based on old recommendations, consumption of refrigerator dill pickles could typically occur
anytime after 3 days of refrigerated storage
- Since L. monocytogenes may still be viable well after this point, there is a food safety risk involved
- Recommendations to prepare this product in the home should not be distributed
This material is based upon work supported by the Cooperative State Research, Education, and Extension Service, U.S. Department of Agriculture, under Agreement No. 00-51110-9762.
Document Use:
Permission is granted to reproduce these materials in whole or in part for educational purposes only (not for profit beyond the cost of reproduction) provided the authors and the University of Georgia receive acknowledgment and this notice is included:
Reprinted with permission of the University of Georgia. Jin Kyung Kim, Elaine M. D'Sa, Mark A. Harrison, Judy A. Harrison and Elizabeth L. Andress. 2004.
Listeria monocytogenes survival in refrigerator dill pickles.
Athens, GA: The University of Georgia, Cooperative Extension Service.
References to commercials products, services, and information is made with the understanding that no discrimination is intended and no endorsement by the University of Georgia, U.S. Department of Agriculture and supporting organizations is implied. This information is provided for the educational information and convenience of the reader.
The University of Georgia and Ft. Valley State University, the U.S. Department of Agriculture and counties of the state cooperating. The Cooperative Extension Service, the University of Georgia College of Agricultural and Environmental Sciences offers educational programs, assistance and materials to all people without regard to race, color, national origin, age, sex or disability. An Equal Opportunity Employer/Affirmative Action Organization Committed to a Diverse Work Force.
Contact:
 =0.05
=0.05 =0.05
=0.05






